Featured Patient Story
Neal McMichael
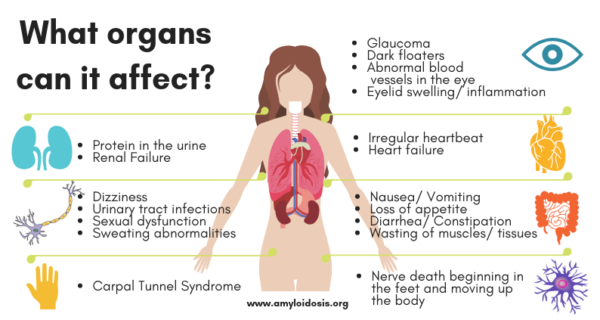
I was diagnosed with Carpal Tunnel Syndrome and had CTS surgery on both my left and right wrists in late 2017. I had my annual physical in January 2018, Blood Testing (TSH) found hypothyroidism (Hashimoto’s) and also found an M-Spike (High Protein Level) and I became extremely fatigued over the next 6-8 months.
read moreLatest News Story Headlines
April 22, 2025 | 5 min read | Nick Paul Taylor
Calendar & Upcoming Events
Webinar: HeartTalks - When Heart Symptoms Don’t Add Up: Understanding ATTR-CM
Webinar: Choosing your Identity and how it impacts the Amyloidosis Experience
Sign Up for the Latest Updates
Get news from Amyloidosis Foundation
in your inbox
By submitting this form, you are consenting to receive marketing emails from : Amyloidosis Foundation, 7151 N. Main St., Suite 2, Clarkston, MI, 48346, US, https://www.amyloidosis.org. You can revoke your consent to receive emails at any time by using SafeUnsubscribe© link, found at the bottom of every email. Emails are serviced by Constant Contract.